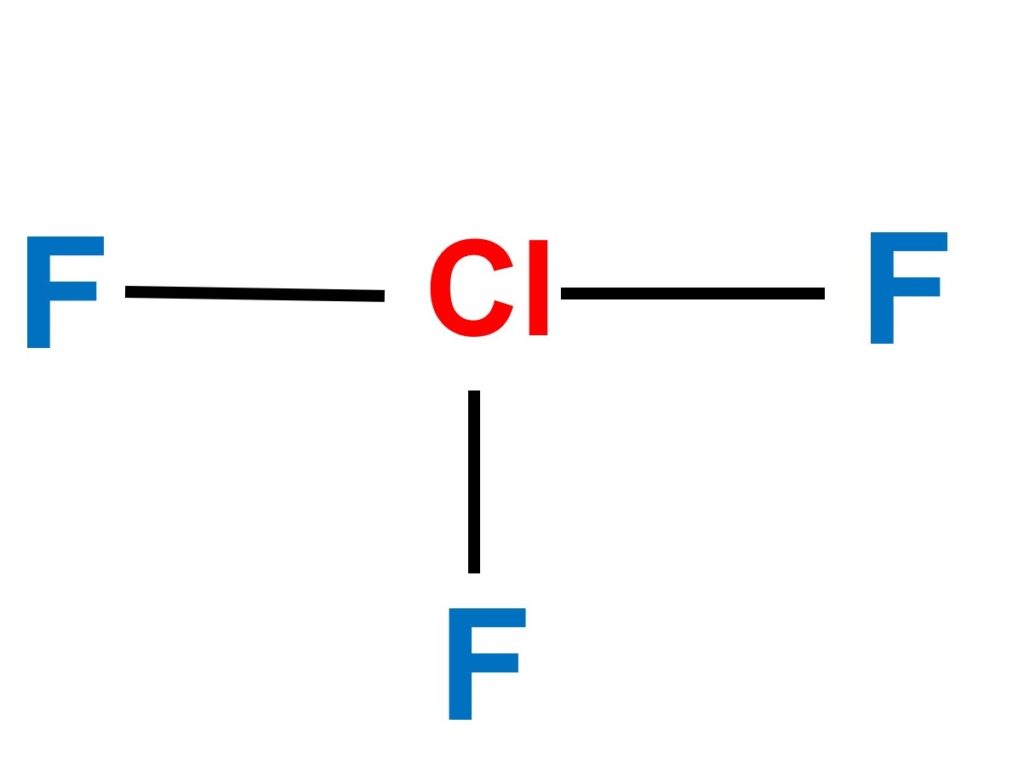
Jacox, Vibrational and Electronic Energy Levels of Polyatomic Transient Molecules, J. Therefore the proportion of carbon in the compound is 36/44, which cancels down to 9/11. The total mass of this that is carbon is 12x3, which is 36. The total mass is 12x3 + 8x1, which equals 44. , Tables of Molecular Vibrational Frequencies, Consolidated Volu i did 9/11 / 12 for carbon and 2/11 / 1 for hydrogen. Maryott "Selected Values of electric dipole moments for molecules in the gas phase" NSRDS-NBS10, 1967 It has a trigonal bi-pyramidal structure. Please address comments about this page to Thakkar, T Wu "How well do static electronic dipole polarizabilities from gas-phase experiments compare with density functional and MP2 computations?" J. This is evident from its molecular structure, which is trigonal bi-pyramidal. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites.įurther, NIST does not endorse any commercial products that may be mentioned on these sites. There may be other web sites that are more appropriate for your purpose. No inferences should be drawn on account of other sites being referenced, or not, from this page. 
We have provided these links to other web sites because they may have information that would be of interest to you. You are here: Experimental > One molecule all propertiesĮxperimental data for ClF 3 (Chlorine trifluoride)Ĭhlorine fluoride Chlorine fluoride (ClF3) Chlorine trifluoride Chlorotrifluoride Trifluorure de chlore īy selecting the following links, you may be leaving NIST webspace.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
